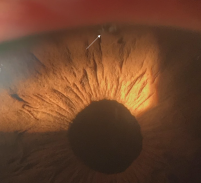
Laser Peripheral Iridotomy
| Primary authors |
|
|---|
WATCH LASER PERIPHERAL IRIDOTOMY: TIPS AND TRICKS: https://www.youtube.com/watch?v=Qzp5MmOPGVo
Laser peripheral iridotomy (LPI) is a laser procedure that creates an aper-ture in the peripheral iris. It is an effective treatment for relieving pupil block in eyes with acute primary angle-closure (APAC) and a prophylactic measure indicated for protecting the fellow eyes from APAC. Prophylactic LPI is also commonly considered in eyes with occludable angles (usually defined as an angle in which at least 270 degrees of the posterior trabecular meshwork can-not be visualized).
Principle
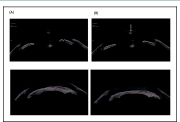
In relative pupil block, there is an increase in resistance to the flow of aqueous humor at the pupillary margin, resulting in a pressure gradient between the posterior and anterior chambers. With a greater pressure poste-rior to the iris, the iris assumes a characteristic forward-bowing appearance (Figure 23-1A). LPI provides a communication between the posterior and anterior chambers, thereby eliminating the pressure gradient, flattening the iris, and opening up the anterior chamber angle (Figure 23-1B).
Instrumentation
LPI can be performed with slit lamp-mounted Nd:YAG laser alone or with sequential argon Nd:YAG laser. A contact lens (eg, Abraham iridotomy lens) is useful to keep the eyelids separated and the globe stable.
Technique
- Perform a complete ophthalmic examination. Review the risks and benefits of the procedure with the patient and obtain a signed informed consent.
- Instill 1 drop of brimonidine 0.15% (to reduce the risk of a post-LPI intraocular pressure [IOP] rise) and pilocarpine 2% (to thin the peripheral iris and widen the angle; Figure 23-2) 30 minutes prior to LPI.
- Instill one drop of topical anesthesia (eg, proparacaine 0.5%).
- Apply a contact lens to the globe and identify a suitable location for laser application. The treatment site is preferably located at the peripheral third of the superior iris from 10 to 2 o’clock position (12 o’clock is usually avoided as gas bubble formation may hamper completion of the procedure) and completely covered by the eyelid (some patients with iridotomy partially covered by the eyelid have reported glare and double vision). In an aphakic eye with silicone oil, LPI is performed at the inferior iris. An area of thin iris or a crypt is preferable as less laser energy is required to penetrate the iris.
- Laser treatment:
- Nd:YAG laser alone: 4 to 6 mJ, 1 to 3 pulses per shot for 1 to 3 applications.
- Sequential argon Nd:YAG laser: (1) Argon laser size: 50 µm,duration: 0.05 to 0.1 second, energy: 700 to 1000 mW. (2) Nd:YAG laser: 1 to 3 mJ, 1 to 2 pulses per shot. The actual laser energy required and the treatment chosen depend on the color and the thickness of the iris. For example, sequential argon Nd:YAG laser may be more effective than Nd:YAG laser alone in dark brown iris with thick stroma; argon laser can minimize bleeding and create a stromal crater for subsequent penetration by Nd:YAG laser application. The size of iridotomy is usually 150 to 200 µm.
- Measure the IOP 1 hour after the procedure to check for IOP rise.
- Topical steroid (eg, dexamethasone 0.1% or prednisolone acetate 1%) is commonly prescribed for 5 to 7 days to reduce intraocular inflammation after the procedure.
Complications
The most common complications following LPI are anterior uveitis and transient IOP elevation. Anterior uveitis is usually self-limiting and resolves in 1 to 2 weeks with topical steroid treatment. A rise in IOP >6 mm Hg 1 to 2 hours after LPI has been reported in 40% of patients with narrow angles or chronic angle-closure glaucoma. One drop of brimonidine 0.15% before or after LPI is often effective in preventing IOP spike. Although lens opacity formation beneath an iridotomy may occur following LPI, published data on cataract progression are conflicting.
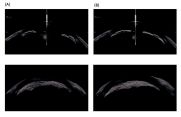
Visual symptoms including ghost images and shadows can occur in up to 4% of patients. The location of the LPI might influence the incidence of visual disturbances. See more information on this topic here: https://www.youtube.com/watch?v=Qzp5MmOPGVo. Placing the LPI superiorly (figure 23-3) at a point intersection by the superior lid margin and/or the superior tear film meniscus is likely to cause defraction of light through the LPI with the patient noticing light crescents or shadows. More temporal (figure 23-4) placement can lead to less visual disturbances but can the procedure is more painful in this location due to the proximate long ciliary nerves.
Hyphema, damage to the corneal endothelium, retinal injury, and malignant glaucoma are other potential but rare complications.

Follow-Up
Although LPI has been proven effective in reducing the risk of APAC, a significant number of patients may still develop chronic elevation of IOP in the presence of a patent iridotomy. Therefore, patients should be monitored for development of peripheral anterior synechiae and angle-closure glaucoma at subsequent follow-up visits.
Suggested Readings
de Silva DJ, Gazzard G, Foster P. Laser iridotomy in dark irides. Br J Ophthalmol. 2007;91(2):222-225.
Drake MV. Neodymium:YAG laser iridotomy. Surv Ophthalmol. 1987;32(3):171-177.
Saw SM, Gazzard G, Friedman DS. Interventions for angle-closure glaucoma: an evidence-based update. Ophthalmology. 2003;110(10):1869-1878.