Neovascular Glaucoma
| Primary authors |
|
|---|
Neovascular Glaucoma
Video Lecture: https://youtu.be/nfXTKDROYBc
Introduction
Neovascular Glaucoma (NVG) presents as a secondary open-angle or secondary closed-angle glaucoma, depending on the extent of fibrovascularization and synechiae formation in the drainage angle. The abnormal vessels form in response to ischemia of the retina and are composed of highly permeable thin walls (lacking a muscular layer or normal adventitia) with absence of tight intercellular junctions. Aqueous outflow is compromised by the fibrovascularization with/without synechiae formation (which form when the fibrovascular membranes, composed of fibroblasts with smooth muscle differentiation, contract over time) leading to increased intraocular pressure (IOP).
Risk Factors
Retinal ischemia is the common denominator leading to NVI/NVA and eventually NVG and can be associated with diabetes, retinal vascular occlusive disease (CRVO and BRVO), tumors, ocular or periocular radiation, Uveitic Diseases and Iatrogenic/surgical causes. In cases involving ischemic CRVO, the NVG has classically been described as “90 day glaucoma” with IOP compromise occurring 90 days after the CRVO event. This is not a firm timeline and glaucoma can occur in these eyes much earlier or later after the initial vascular event.
Diagnosis and Clinical Course
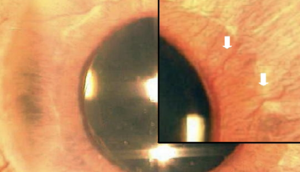
Diagnosis of NVG is based on clinical examination and can be assisted by fluorescein angiography. The first sign of new vessel growth in the anterior segment is leakage of intravenously injected fluorescein dye from vessels at the pupillary margin. This is the pre-rubeosis stage as NVI/NVA are not yet detectable. When vascular proliferation occurs, signifying transition to the rubeosis phase, it is observable by slit-lamp biomicroscopy typically at the pupillary margin (Figure 1). When NVI is observed, careful gonioscopy is necessary to identify any NVA and to quantify any existing fibrovascular membranes or peripheral anterior synechiae. In fact, any patient at risk of developing NVG should undergo periodic inspection of the anterior chamber angle by gonioscopy since findings in the angle might precede slit lamp findings on the pupillary margin or iris stroma (as can be the case especially in patients with dark irides). NVI can be associated with Ectropion Uveae, which is the presence of iris pigment epithelium on the anterior surface of the iris, in later stages. Ectropion Uveae occurs in NVG when the pupil margin is pulled by the abnormal vascular and fibrovascular membranes causing a folding at the margin with anteriorization of the posterior surface of the iris.
As vessels proliferate in the angle, arborizing over the ciliary body and scleral spur and spreading over the trabecular meshwork (TM), progressive elevation in IOP can be noted related to a decrease in outflow facility. NVA typically occurs first at the base of the iris taking the form of individual vascular trunks that cross the ciliary body and scleral spur and over the corneoscleral TM. The vessels then form fine networks of branching connections that become intertwined with branches from other vascular trunks. Eventually, a fibrovascular membrane forms that is contractile and produces localized anterior synechiae (which do not reach beyond Schwalbe’s line as can be seen in ICE Syndrome) with progressive zipping of the angle as the disease progresses. The elevation of IOP at these later stages can occur acutely with extremely high applanation values and red painful eyes.
Differential Diagnosis
Acute or Chronic Uveitis, Acute or Chronic Angle Closure Glaucoma, Ocular Tumors, Iatrogenic (post-surgical IOP spikes), Trauma.
Anterior segment neovascularization can occur without retinal ischemia as is the case with pseudoexfoliation syndrome, isolated iris melanomas, Fuchs heterochromic uveitis and other types of uveitis.
Treatment
Topical and oral IOP lowering medications are temporizing measures and surgery is almost always required since presentation is often at stages where the IOP is elevated and the drainage angle is compromised with significant “zipping” of the aqueous outflow system. Anti-VEGF agents should be injected into the vitreous cavity or anterior chamber (both are acceptable to address the NVA/NVI) and scheduling of surgery to reduce IOP typically involves prompt placement of a glaucoma drainage device. The anti-VEGF agents can cause reversal of neovascularization and decrease the chance for intraoperative bleeding from friable vessels along with decreasing the pro-inflammatory angiogenic drive. In cases that are less urgent, pan-retinal photocoagulation (PRP) can be scheduled to treat the ischemic retina and decrease the angiogenic drive from compromised tissues. Regardless of when PRP is done (before or after addressing the IOP elevation), laser is often needed to treat retinal pathologies since chronic injections of anti-VEGF agents is less desirable and less efficient. The glaucoma drainage device of choice is typically the Ahmed Valve (FP7, New World Medical) since IOP is usually extremely elevated and prompt lowering of pressure is needed (best done with a valved device). Trabeculectomy surgery has a very high failure rate in these eyes and is not considered standard of care unless the NVG has been treated with PRP/Anti-VEGF agents and the disease process has been controlled for some time prior to filtration surgery. Novel devices such as MIGS implants or Goniotomy are not appropriate in these eyes due to angle pathology and need for significant lowering of IOP. Finally, cyclophotocoagulation (transscleral or endoscopic) can be utilized if GDDs are not a good option but can lead to significant morbidity and hypotony with poor ability to titrate therapy given the variable aqueous inflow and outflow compromise in NVG.
Further Reading:
Wand M., Neovascular glaucoma. In: Ritch R, Shields MB, Krupin T, eds. The Glaucomas – Clinical Science. 2nd. Ed. St. Louis: Mosby; 1996: 1073-1129. Ch 51.
Hayreh SS., Neovascular glaucoma. Prog Retin Eye Res. 2007; 26(5): 470-485.
Kahook MY, Schuman JS, Noecker RJ. Intravitreal bevacizumab in a patient with neovascular glaucoma. Ophthalmic Surg Lasers Imaging. 2006 Mar-Apr;37(2):144-6.