Congenital and Pediatric Glaucoma: Goniotomy and Trabeculotomy
| Primary authors |
|
|---|
Goniotomy and trabeculotomy are performed to improve the facility of outflow of aqueous humor from the eye by incising abnormal trabecular meshwork. This creates an improved conduit between the anterior chamber and Schlemm’s canal rather than creating an alternative aqueous bypass as in filtration or tube shunt surgery. This chapter focuses on the pediatric applications of these two procedures.
Examination Under Anesthesia
Examination under anesthesia contributes essential information in preparation for angle surgery and is very often necessary in caring for pediatric patients. To minimize artificially low intraocular pressure (IOP) measurements, which can be caused by inhaled anesthetics, IOP measurements should be taken soon after the induction of anesthesia. IOP measured during an examination under anesthesia must be considered in conjunction with the other findings because the pressure measured is often different than that measured if the child were awake. A Tonopen (Reichert technologies, Depew, New York), Schiotz, or Perkins tonometer is used. Calipers or a ruler is used to measure the corneal diameter for future comparison. Axial length measurements are also helpful for future comparison and can be used in concert with changing refractive error and corneal diameter to identify disease progression. Careful slit-lamp examination should then be performed. A diagnostic goniolens such as a Koeppe lens coupled with balanced salt solution is then placed on the cornea, and a hand-held slit-lamp device can be used to carefully examine the cornea, iris, and angle structures. Alternatively, a mirrored lens or a Koeppe lens may be used with the operating microscope. Both the cornea and angle structures can be examined while the goniolens is in place. The examiner should look for evidence of Haab’s striae, which are horizontal breaks in Descemet’s membrane. In addition, corneal stromal and/or epithelial edema may be noted. The quality of the view to the angle is crucial in deciding between the goniotomy and trabeculotomy procedures. The surgeon must evaluate the angle and become familiar with its anatomy. Careful observation will reveal abnormalities in the angle and iris structures, as well as evidence of previous angle procedures. This information is helpful not only in determining the etiology of glaucoma but also to orient the surgeon with the architecture of the angle prior to the procedure. Finally, a direct ophthalmoscope may be used to examine the optic nerve through an undilated pupil. This can also be accomplished through the pupil with the Koeppe lens in place after completing gonioscopy.
Goniotomy
Indications
Goniotomy is the procedure of choice for infantile primary congenital glaucoma and is also used for other types of developmental glaucoma, as well as some types of secondary glaucoma. However, angle surgery is not indicated in all types of childhood glaucoma. The decision about type of surgery has to be tailored to the individual’s diagnosis and case. Goniotomy and trabeculotomy obtain similar results in experienced hands; however, individual surgeons may have a preference for one procedure over the other. Trabeculotomy is particularly helpful when the angle view is limited by opacification of the cornea. Advantages of goniotomy include a relatively short procedure time and preservation of conjunctiva for future filtration procedures. Also, anatomical landmarks for a trabeculotomy may be challenging to identify in a severely buphthalmic eye.
Preparation and Instrumentation
Glaucoma medications, such as oral acetazolamide 10 to 15mg/kg/day, may be used preoperatively to help reduce the IOP, as well as to help clear the cornea, so that a goniotomy may be performed. Other topical glaucoma medications may also be used preoperatively to help reduce pressure, including topical carbonic anhydrase inhibitors and beta-blockers. Alpha-adrenergic receptor agonists are typically avoided in infants because of possible somnolence and apnea. The exception to this is the intraoperative use of apraclonidine 0.5% (see Surgical Procedure section).
Goniotomy
|
Trabeculotomy
|
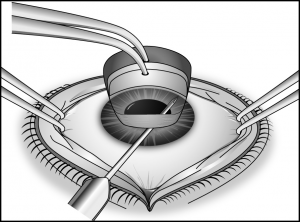
Goniotomy may be performed with an operating microscope or with loupes for magnification. The operating microscope is tilted to 45 degrees to provide an optimal view of the angle structures. During the cleaning and preparation, a few drops of dilute betadine are placed in the eye. The surgeon is seated opposite the operative angle. A trained assistant can help to stabilize and rotate the eye during the procedure. See Table 30-1 for appropriate surgical instruments and supplies.
Surgical Procedure
Typically, goniotomy is performed under general anesthesia. First, examination under anesthesia is performed as described above. The cor-neal epithelium may be removed if edematous using 70% isopropyl alcohol to improve visualization. Apraclonidine 0.5% on a spear (to limit the amount of medication absorbed systemically and avoid mydriasis) may be applied to the limbus adjacent to the planned goniotomy cleft to reduce the reflux of blood into the anterior chamber. The surgeon sits opposite the portion of the angle to be operated with the patient’s head turned slightly away from the surgeon. The assistant stabilizes the eye with locking forceps secured at the insertion of the superior and inferior rectus muscles. Next, the operating goniolens is placed on the cornea.
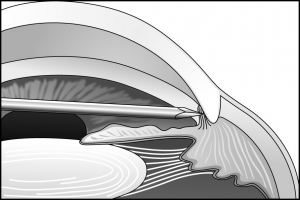
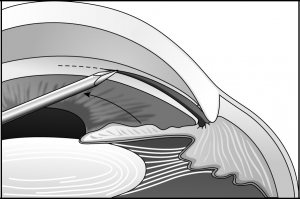
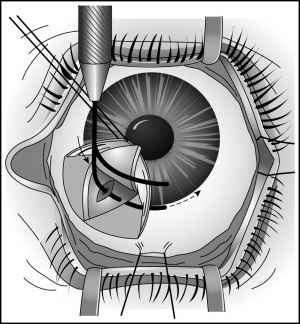
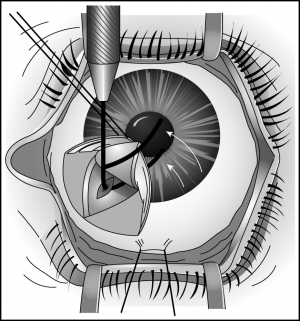
Either a goniotomy knife or a 25-gauge needle on a viscoelastic-filled syringe is introduced into the anterior chamber through the peripheral cor-nea. The instrument’s tip is visualized as it is advanced across the anterior chamber, parallel to the iris, to the opposite angle (Figure 30-1). It is then introduced to the mid to posterior third of the trabecular meshwork and a circumferential incision is made in one direction and then in the opposite direction, with the assistant carefully rotating the eye when requested by the surgeon (Figures 30-2 and 30-3). During the procedure, the surgeon will note the iris moving posteriorly, the release of iris processes, and the formation of a cleft. Approximately 4 clock hours can be treated with one procedure. After this incision is made, the instrument is removed from the anterior chamber and the anterior chamber is deepened with balanced salt solution or a sterile gas bubble. Normally, reflux of blood into the anterior chamber will occur concurrent with shallowing of the anterior chamber and hypotony. After the instrument is removed, forceps points held together are used to gently hold the incision closed. A 10-0 absorbable suture is used to secure the incision with the knot buried. The eye is then dressed with topical steroid drops and antibiotic drops. A patch and shield are then placed on the eye.
Complications and Prognosis
A small hyphema is common following goniotomy and typically clears within a few days. Observe the patient closely for progression of hyphema, which may require an anterior chamber washout. More severe trauma to the iris and angle structures, such as iridodyalysis or cyclodyalysis, can also occur, potentially leading to hypotony. Trauma to the cornea and retinal detachment can also occur. Great care should be taken to avoid trauma to
the lens and subsequent cataract formation. Corneal edema can obscure the surgeon’s view and increase the risk of these complications. If a tapered knife is used, it is important to be sure that the chamber does not collapse, which could lead to lens trauma. There is also a small risk of intraocular infection or inflammation. Prognosis is dependent on the etiology of glaucoma. Many children require further surgical intervention to control IOP.
Key Points of Goniotomy Surgery
- Proper positioning of the patient, microscope, and assistant are crucial to performing a successful goniotomy. Practicing the procedure with the assistant just before the procedure can alert the team to potential difficulties that may arise during the procedure. For example, the surgeon’s view may not be adequate or the assistant may be unaware of how to manipulate the eye smoothly in a given position.
- It is easy to introduce air bubbles under the goniolens when placing it on the eye. To avoid this, the head of the patient is tilted away from the surgeon; the lens is initially placed at a 45-degree angle to the cornea as the goniolens is lowered to the surface of the cornea over a small amount of saline or viscoelastic.
- A gonioknife typically has a tapered shaft. As a result, it is possible for the corneal wound to leak as the blade is removed from the anterior chamber. This is not true for a 25-gauge needle, which has a straight shaft. The needle is placed on a viscoelastic-filled syringe and the chamber can be deepened if it becomes shallow during the procedure.
- If a 25-gauge needle is used for the procedure, care should be taken to enter the cornea parallel to the iris so that the flexible needle does not pivot in an anterior-posterior direction while the angle incision is being created.
Trabeculotomy
Indications
Trabeculotomy is a surgical procedure that can be performed when the cornea is not adequately clear to view the angle structures and perform goni-otomy. Some surgeons choose trabeculotomy as their procedure of choice when angle surgery is indicated, particularly if performing a 360-degree suture trabeculotomy is preferred. A 360-degree suture trabeculotomy offers the potential to treat the entire angle with one procedure. Angle surgery is often the procedure of choice for a child with glaucoma but not for all forms of glaucoma. The decision about type of surgery has to be tailored to the individual’s diagnosis and case.
Preparation and Instrumentation
Following the examination under anesthesia, the eye is prepared and positioned based on the surgeon’s preferred approach. A temporal approach is often selected to avoid the nose and to preserve the superior conjunctiva if future filtration surgery is needed. During the cleaning and preparation, a few drops of dilute betadine are placed in the eye. See Table 30-1 for appropriate surgical instruments and supplies.
Surgical Procedure
Conjunctival Peritomy and Scleral Flap Creation
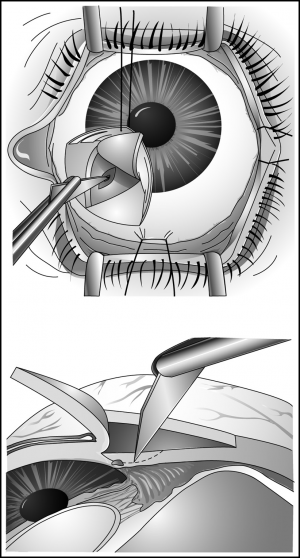
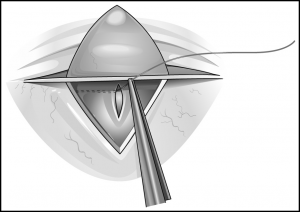
A fornix-based peritomy for approximately 3 clock hours is performed with Westcott scissors and fine forceps to expose sclera above Schlemm’s canal for the creation of a 3-mm equilateral triangular scleral flap (Figure 30-4). Light cautery can be used to achieve hemostasis. The scleral flap is partial thickness and created with the #57 blade. Dissection of this flap is extended anteriorly until darker limbal tissue is easily seen anterior and adjacent to the sclera.
Dissection to Schlemm’s Canal
A 2-mm radial incision is created with the #57 blade at the junction of the scleral and limbal tissue (see Figure 30-4). Successive deeper layers of sclera are dissected by “scratching down” with gentle downward pressure and side-to-side movements of the #57 blade. The circumferential fibers of Schlemm’s canal may be appreciated on dissection to the outer wall of the canal. The use of higher magnification and a sharp pointed blade can facilitate this final dissection. Entry into Schlemm’s canal is heralded by a slow egress of blood-tinged aqueous humor. Rapid egress of fluid should raise suspicion of inadvertent direct entry into the anterior chamber. If inappropriate entry into the anterior chamber has occurred, the incision should be sutured closed with 10-0 absorbable sutures and the dissection should be performed slightly posterior to the initial site. The presence and patency of Schlemm’s canal may be confirmed by introducing a short segment of 6-0 Prolene or nylon suture material, which should enter with minimal difficulty. If significant resistance is encountered, consider which anatomic space has been entered (eg, the suprachoroidal space). Once Schlemm’s canal has been successfully identified, a peripheral corneal paracentesis is created to allow reformation of the anterior chamber with ophthalmic viscoelastic devices if needed.
Opening of Trabecular Meshwork
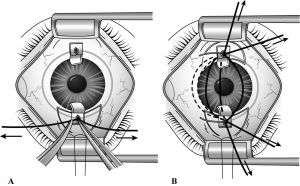
Using the metal Trabeculotome, the distal arm of the Trabeculotome is placed through Schlemm’s canal at the dissection site. The proximal arm is used as a guide and should track along the limbus external to the eye as the distal arm moves with minimal resistance through Schlemm’s canal circumferentially for approximately 2 hours (Figure 30-5). The instrument is then rotated into the anterior chamber over the iris leaf surface until three-fourths of the length of the Trabeculotome is directly visualized in the anterior chamber (Figure 30-6). The Trabeculotome is then withdrawn.
Often the anterior chamber shallows and/or hyphema occurs. Injecting an ophthalmic viscoelastic device (OVD) through the paracentesis may be needed to partially reform the anterior chamber and move the iris posterior before initiating use of the opposite metal Trabeculotome. The opposite Trabeculotome is placed through the opposite side of the incision into Sch-lemm’s canal for an additional 2 clock hours and rotated into the anterior chamber as previously described.
If suture trabeculotomy is chosen instead of the metal Trabeculo-tome, a 6-0 Prolene suture is measured along the external border of the limbus and trimmed to a few millimeters longer than this measured distance. The leading end of the suture is melted into a mushroom shape with the use of cautery held in close proximity to the suture without contact. This mushroomed leading end of the suture is thread through Schlemm’s canal and recovered from the opposite side of the incision after passing 360 degrees (Figure 30-7). Resistance to passing the suture may be encountered 180 degrees from the introduction site. If this occurs and it is not easy to pass, a second flap can be created at this location to recover the suture and introduce an additional suture for the remaining 6 clock hours. The leading end is grasped external to the eye, and then both leading and trailing ends are grasped with equal force and pulled tangentially through the trabecular meshwork into the anterior chamber (Figure 30-8).
Closure
The scleral flap is closed with interrupted 10-0 absorbable sutures; typically a single suture is sufficient. If significant OVD or hyphema is in the anterior chamber, the flap can be loosely closed and serve as a temporary
filter. Finally, the peritomy is closed watertight with 10-0 absorbable suture. After applying antibiotic and corticosteroid medications, the eye is patched and shielded.
Complications and Prognosis
False passages are possible, either into the anterior chamber or the suprachoroidal space. Postoperative hypotony is therefore a risk. Postoperative hyphema often occurs, but viscoelastic and apraclonidine 0.5% can help to control this. Observe the patient closely for progression of hyphema, which may require an anterior chamber washout. A small risk of postoperative infection exists. The patient needs to be followed closely for an elevation of IOP; repeat angle surgery or other procedures and medication control may be required. Prognosis is often related to the etiology of the glaucoma.
Key Points of Trabeculotomy Surgery
- Preparing the correct location and positioning the patient prior to entry into the eye is an important first step of a successful procedure.
- A partial-thickness scleral flap allows for easier identification and dis-section to Schlemm’s canal.
- A gentle approach to Schlemm’s canal is used by scratching down at the scleral–limbal junction through the scleral fibers with a knife in a side-to-side motion with slight downward pressure.
- When Schlemm’s canal is located, slow egress of blood or pigment-tinged fluid will be appreciated. Rapid egress of fluid should raise suspicion of entry into the anterior chamber.
- Entering the suprachoroidal space with either the suture or metal Trabeculotome is possible. Significant resistance to passing the instrument should raise suspicion for the incorrect location.
- With initial rotation of the metal Trabeculotome, if it is directed posteriorly, movement of the iris will be seen. If this position is not corrected, an iridodialysis will occur.
- To avoid corneal or lenticular damage, OVDs may be necessary to maintain and deepen the anterior chamber partway through the procedure.
Suggested Readings
Beck AD, Lynch MD. 360-degree trabeculotomy for primary congenital glaucoma. Arch Ophthalmol. 1995;113(9):1200-1202.
Freedman SF. Medical and surgical treatments for childhood glaucomas. In: Allingham RR, ed. Shields’ Textbook of Glaucoma. 5th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:626-643.
Mandal AK, Netland PA. The Pediatric Glaucomas. Philadelphia, PA: Elsevier; 2006.
Walton DS. Goniotomy, trabeculotomy, and goniosynechiolysis. In: Higginbotham EJ, Lee DA, ed. Clinical Guide to Glaucoma Management. Woburn, MA: Butterworth Heinemann; 2004:412-423.
Walton DS. Pediatric glaucoma: angle surgery and glaucoma brainage devices. In: Giaconi JA, Law SK, Coleman AL, Caprioli J, eds. Pearls of Glaucoma Management. Berlin, Ger-many: Springer-Verlag; 2010:403-408. </references>