Traumatic Glaucoma
| Primary authors |
|
|---|
Traumatic Glaucoma
Video Lecture: https://youtu.be/yBjWiXvHIv8
Traumatic glaucoma is any form of glaucoma that results directly from injury to the eye. Some causes include blunt trauma with direct damage to the aqueous outflow system, penetrating injuries leading to disruption of tissues with inflammation and scar formation, retinal detachment with subsequent plugging of the trabecular meshwork with blood or inflammatory cells (or photoreceptors), bleeding with compromise of the outflow system as well as exposure to caustic substances. The elevation in intraocular pressure (IOP) can occur immediately after trauma (bleeding) or decades later (angle recession). The risk of development of glaucoma post blunt trauma has been reported at 3.39% and 2.67% after penetrating injuries.
Causes of IOP Rise Post Ocular Trauma:
•Persistent inflammation is often temporary post trauma but can be chronic with resulting changes to the drainage angle (inflammatory cell deposition and/or synechiae formation) that can lead to lasting elevation in IOP and development of glaucomatous optic neuropathy (GON). Strict control of inflammation with steroids can decrease the chance of synechial angle closure but must be closely supervised to avoid steroid related IOP spikes that can in itself cause GON. Extensive synechiae formation may necessitate goniosynchialysis (https://youtu.be/d5YgdWgRU44) once inflammation is controlled to allow for egress of aqueous humor out of the eye. Other surgical procedures may also be necessary depending on the stage and severity of disease.
•Hyphema is another cause of IOP elevation post trauma to the eye and requires prompt attention to avoid long-term complications. The initial hyphema often resolves with use of steroid drops along with mydriatics (Atropine 1%) but resolution depends on the extent of the hyphema. A small layered 1-2 mm hyphema is initially less concerning relatively speaking compared to an 8 ball hyphema that can lead to immediate elevation of IOP and staining of the cornea if longer lasting. However, even small layered hyphemas can be associated with significant IOP spikes and may even require surgical intervention in the near term depending on the response to therapy as well as the baseline status of the optic nerve and how resilient it may be in the face of spiking IOP. IOP lowering medications should be utilized to control pressure and most classes of topical drops are appropriate (PGA, BB, AA, CAI) although miotics should be avoided due to possible increased inflammation (with compromise of the blood-eye barrier) as well as the potential to alter the anatomy of the angle with breaking of clots and exacerbation of traumatic effects on the AH outflow system. One major concern is when an initial hyphema is resolving but a rebleed occurs:
•5-10% rebleed with increased risk of glaucoma
•Rebleed occurs at 3-7 days due to clot retraction/lysis
•High IOP with hyphema: corneal staining (time and IOP dependent)
•Sickle cell: RBCs plug TM (low pH) and ONH at higher risk
•HOB elevate, patch, steroids, Atropine, AH suppressants
•Avoid CAI in patients with Sickle Cell, avoid Pilocarpine in all.
•AC washout +/- filtration surgery may be necessary (see below for details on AC washout that we use in our academic practice).
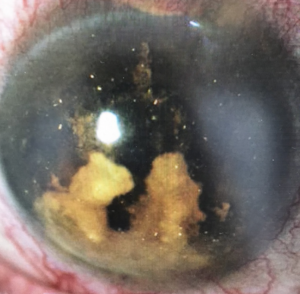
•Cholesterolosis post hyphema: This is a relatively rare finding of cholesterol crystals in the anterior chamber of an eye (Figure 1) with history of blunt or surgical trauma
and hyphema (often extensive and long lasting, but not always). The crystals form due to a rise in insoluble cholesterol concentrations within ocular fluids and is not
related to level of cholesterol in the blood stream.
•Angle Recession v. Ciliary Body Cleft (More information on each here: https://www.keogt.com/Angle_Recession_Glaucoma and here: https://www.keogt.com/Repair_of_Cyclodialysis_Clefts) with emphasis on the need to perform gonioscopy to establish the presence of pathology.
•Lens Subluxation may lead to pupillary block and or peripheral angle closure due to compression of the iris against the cornea. Surgical intervention is needed in these cases with removal of the lens (often combined with vitrectomy) and implantation of an ACIOL or suturing an IOL to the sclera or iris using various techniques.
•Retained foreign body (siderosis, chalcosis) may lead to toxicity with subsequent compromise of the AH outflow system. With siderosis, Iron may accumulate in the trabecular meshwork causing toxicity and cell death. Chalcosis (from copper foreign bodies) can lead to severe inflammation and necrosis with widespread ocular damage including development of glaucoma.
•Chemical burns (alkali>acidic) can both lead to compromise of the outflow system and lifetime monitoring of these eyes is necessary to maintain surveillance of IOP and the optic nerve. Alkali exposure tends to cause worse intraocular pathology due to enhanced penetration into the eye (due to lipophilicity of these agents) compared to acidic exposure (which denature proteins creating barriers to further damage in the eye).
•Choroidal hemorrhage may lead to elevation in IOP both due to the volume effect of blood accumulating in the eyes as well as disruption of outflow of aqueous humor with shallowing of the anterior chamber and potential synechial closure without intervention to treat the condition.
•Schwartz Syndrome: liberated photoreceptor outer segments causing elevated IOP by plug TM with increased IOP post RD (traumatic or otherwise).
Any patient presenting with blunt trauma to the eyes resulting in angle pathology with or without hyphema should be educated about the lifelong increased risk of developing glaucoma and encouraged to keep regular yearly follow up appointments by an eyecare professional. The counseling of patients about this recommendation should be noted in the medical record for future reference.
Tips and Tricks for Successful AC Washout
In the Setting of Ocular Trauma
Deidre St. Peter, MD, Leonard K. Seibold, MD, Malik Y. Kahook, MD
Glaucoma Service
Sue Anschutz Rodgers Eye Center
University of Colorado School of Medicine
Aurora, CO
Pre-operative planning
1) Always look for phacodonesis or lens subluxation at time of pre-operative assessments. Gentle B scan if needed can be helpful when possible. A large differential in anterior chamber depth on UBM between upright and supine positioning may indicate zonular compromise.
2) Caution with use of Pilocarpine (sometimes considered for protection of the crystalline lens prior to surgery) which may lead to shallowing of AC, destabilize clots and increase risk of re-bleed.
3) If concern for lens subluxation or vitreous prolapse, consider combined case with Retina Service.
Anesthesia
1) Consider general anesthesia for some patients (poorly cooperative or when anticipating a long surgical procedure).
2) If foregoing general anesthesia, strongly consider retrobulbar or peribulbar block for pain control.
3) Consider consenting for possible lens removal (in phakic patients) and vitrectomy as well.
Intra-operative considerations
1) Always have phacoemulsification machine with I/A and anterior vitrectomy packet in the room.
2) Consider initially flushing heme through a single paracentesis with BSS to avoid more invasive approach. It is often more efficient to create two paracenteses with one for injection of BSS and one for “burping” to allow for heme to efflux. Creating two paracenteses may also assist with other maneuvers as noted in #13 below.
3) During wound construction, consider longer, angled wounds to prevent iris prolapse.
4) Avoid temporal wounds in case lens capsule is violated and phacoemulsification is needed.
5) Consider using Superepi or epi-shugarcaine to decrease bleeding risk and stabilize iris.
6) Can use manual I/A, Simcoe, or vitrectomy instrumentation for the procedure.
7) Consider coating the endothelium with dispersive viscoelastic for protection.
8) Assume zonular dehiscence and avoid large fluctuations in AC volume. Consider AC maintainer, if needed. Elevate bottle height to deepen/stabilize AC especially if phakic. Lower aspiration to ensure AC stays deep.
9) Do not attempt complete extraction of clot as this may cause rebleed and may injure adjacent structures (iris).
10) The anterior vitrector can be used to trim clots in the angle as much as possible without pulling that may result in re-bleed.
11) In rare instances, diathermy with a 23-guage endodiathermy needle may be attempted if bleeding site visible intraoperatively.
12) One can also consider leaving an air bubble in the anterior chamber at the end of the case, if the source of bleeding is superior, to help tamponade the hemorrhage.
13) If iris prolapses from wounds during procedure:
a. Resist the urge to reposit the iris. Release trapped fluid or viscoelastic from another paracentesis to decompress the AC.
b. After you have lowered the pressure through the second paracentesis, consider stroking the cornea overlying your wound to help release the iris.
c. Can consider making additional paracentesis 180 degrees away to sweep iris from wound.
d. Avoid using dispersive viscoelastic to reposit the prolapsed tissue as this may coat the iris and make it more likely to prolapse.
e. Stabilize the fluidics by lowering the intraocular pressure/ bottle height or decreasing the aspiration flow rate.
14) Always suture wounds in these cases.
References
1. Devgan, U., MD (Producer). (2019, December 24). How to surgically evacuate this hyphema (ocular trauma with hemorrhage) [Video file]. Retrieved October 25, 2020, from https://www.youtube.com/watch?v=Msf3LxX5bJo
2. Surgical Therapy. (n.d.). Retrieved October 25, 2020, from https://www.aao.org/focalpointssnippetdetail.aspx?id=b8b6869e-76db-484d-aeeb52a76051ecb1
3. Christiansen, S. M., MD, & Oetting, T. A., MD. (2017, May 3). Ten Tips to Prevent and Treat Iris Prolapse. Retrieved October 25, 2020, from https://webeye.ophth.uiowa.edu/eyeforum/tutorials/Ten-Tips-Iris-Prolapse.htm
Further Reading:
Yanoff, Myron, MD, Duker, Jay S., MD. Ophthalmology, Fifth Edition.Philadelphia, Pennsylvania: Elsevier Inc; 2019:1089-1094. Osman EA. Glaucoma after open globe injury. Saudi J Ophthalmol. 2015 Jul-Sep;29(3):222-4.